More recent work on this pathway elucidates the complex dynamic patterns of p53 activities that cells use to encode and transmit information. Instead, differences in pulse cycle between cells created a population average behavior showing damped oscillations. However, single-cell studies revealed that there are no damped oscillations in the individual cell. For example, earlier studies of the p53 signaling pathway provided support for damped oscillations to stimulus.

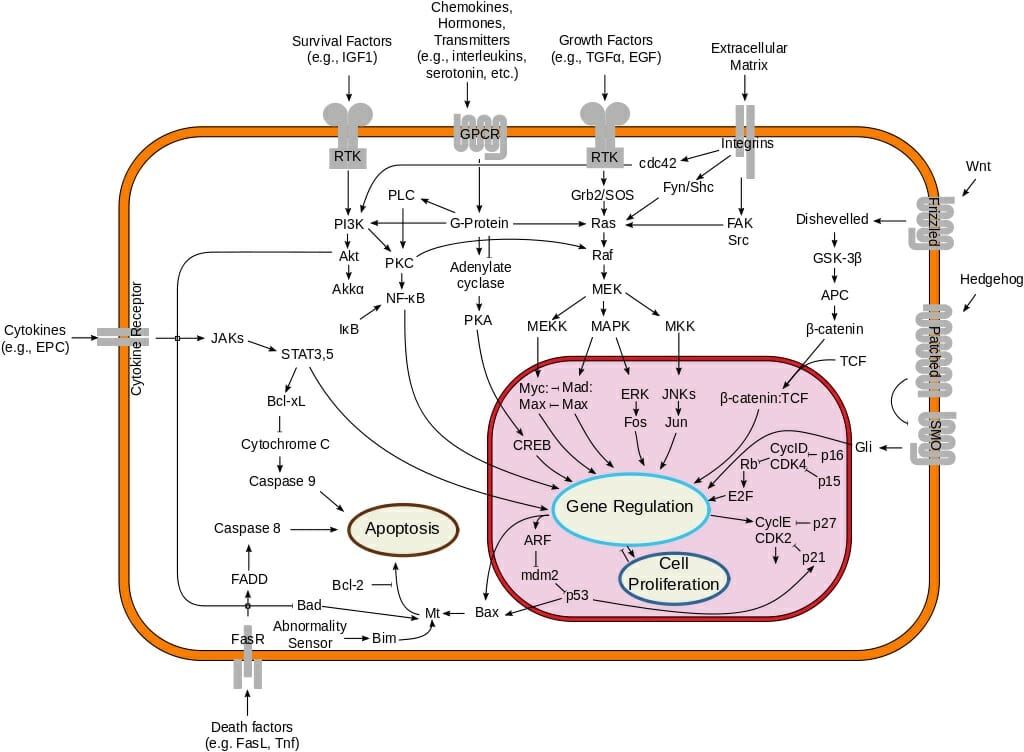
These new studies often reveal that previous knowledge about signaling dynamics at the population level may not be the complete picture. Over the last few years, technological advancements in measuring, manipulating, and modeling signaling activities at single-cell resolution has enabled researchers to go beyond the limitations of population-averaged bulk assays. Furthermore, population averaging results in information loss regarding biological variability that often has important physiological implications. Although these studies have been useful in mapping signaling pathways and making qualitative predictions, population averaging can often mask the spatial and temporal dynamics important in physiological processes. Traditional biological approaches to study cellular signal transduction include bulk assays to measure cellular response at the population level. Understanding the impact of these activating mutations on kit signaling is important, not only in contributing to the understanding of the pathogenesis of mastocytosis but ultimately in forming the basis for more effective therapeutic intervention in this disease.Defining cellular signaling pathways is important to understand many biological processes including tissue development, immune response, cancer development, cellular growth and migration, and more. There is also emerging evidence that inherent inhibitory factors may exist in the juxtamembrane of kit and may be suppressed as a result of a mutation in that region. For instance, this activation could occur through degradation of SHP-1, the protein tyrosine phosphatase that negatively regulates kit signaling. There is, however, mounting evidence that an activating mutation may enhance kit signaling and also induce factor-independent activation of kit. There is still limited information about the effect of activating mutations on various aspects of kit signaling. A signaling network with multiple intersecting pathways can obtain a single, coherent response from numerous, potentially conflicting signals. Ras thus binds PI3 kinase and potentiates its activation, whereas the Rac-dependent protein kinase PAK phosphorylates MEK and thereby stabilizes its association with Raf. There are also numerous cross-connections between signaling proteins more distal to the receptor. Cytoplasmic signaling proteins are increasingly seen to form networks directed through converging and interacting pathways rather than following a simple linear model. The current understanding of kit signaling is that a limited number of signaling proteins interact to build multiple interacting networks that allow diverse cellular responses.
